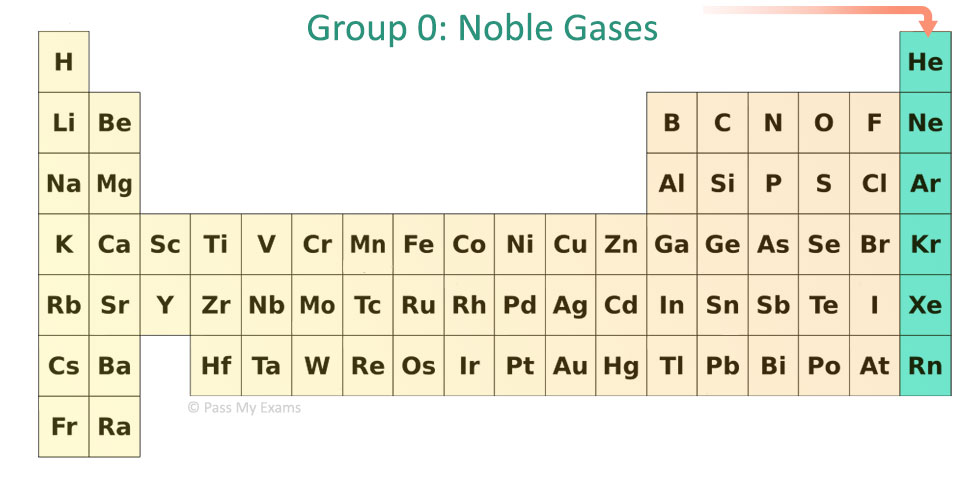
The elements in row 7 are the most reactive, but technically. For example, Te, or Tellurium, is in group 16 and row 5. Reactivity increases as one goes down a group (column), especially in the alkali. Key groups include alkali metals, alkaline earth. The table is divided into metals, nonmetals, and metalloids, each with distinct properties. 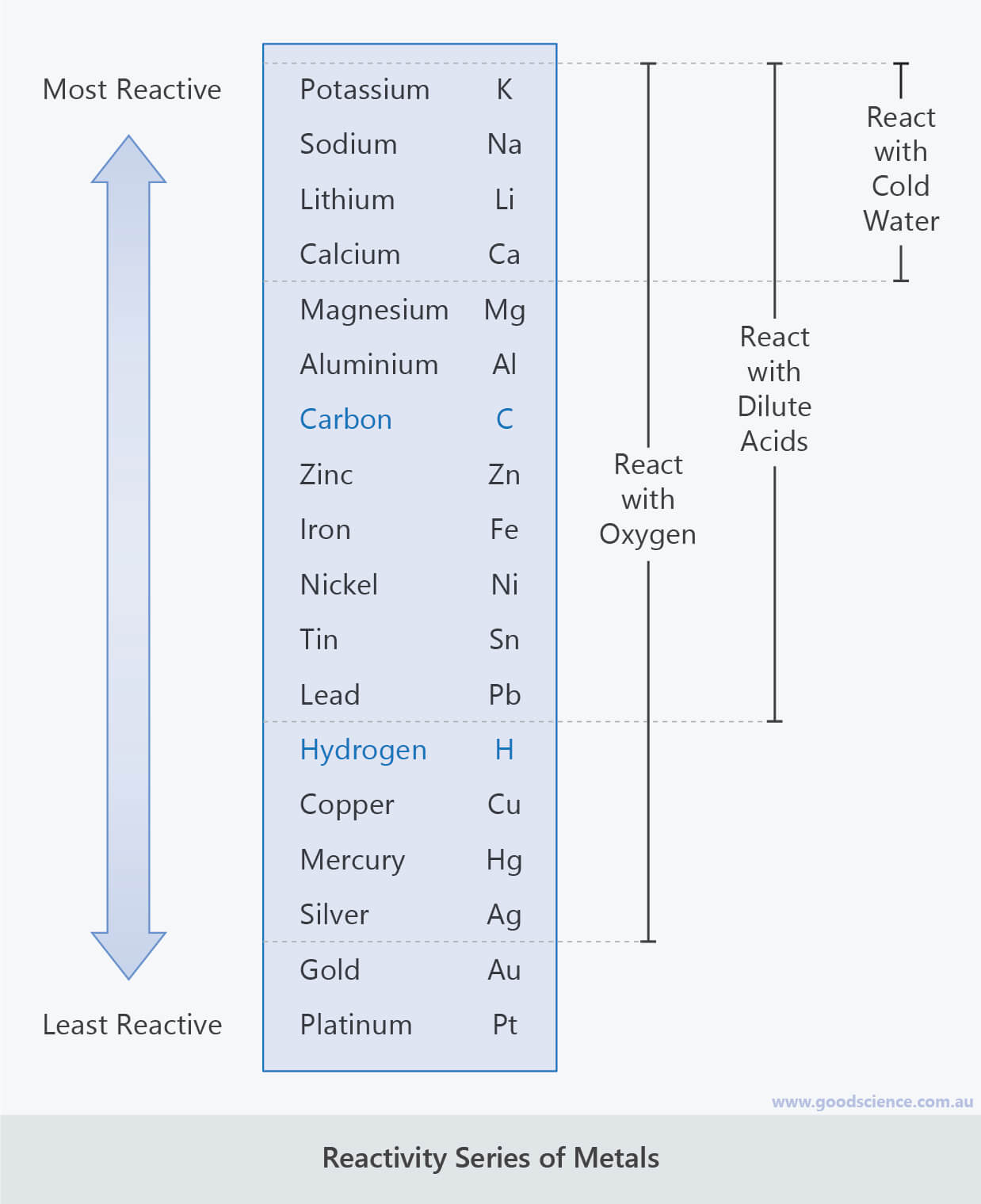
Elements in the same group share similar characteristics, like reactivity. Group 7A (or 17) elements are also called halogens. The more reactive elements on the Periodic Table are farther down in the rows. The two most reactive elements in group 1 are Caesium and Francium. The periodic table organizes elements into groups and periods based on their chemical and physical properties. Trending Questions Sugar allows osmosis in potatoes? What are the chances of getting pregnant from 'pre-ejaculatory fluid'? What color tube used for Triglycerides? How would you classify the following material milk? Is H2O a metal non metal or metalloid? Who invented bottled water and why? What is an atom or group of atoms that has become electrically changed? Is CL from 2NE1 knock-kneed? What does JC on inside of ring mean? How much does francium cost for 1 gram? How does water have a vital role in the creation and breakdown of disaccharides? Why boron trifluoride have maximum covalency is 4? What is the independent variable in rainbow fire experiment? Distilled pure water has a pH of 7 what is it classified as? Where is dihydrogen monoxide Found? Are buffalo nickels made of silver? What is the solubility of marble in normal rainwater for which pH5. It is important to recognize a couple of other important groups on the periodic table by their group name.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
